|
Goldstein postulated that he excluded that the beams could be particles of matter charged with electricity and launched at high speeds because he believed that two beams of canal rays could cross each other without getting in the way and because they did not seem to be influenced by electric or magnetic fields. This curious name should have been temporary, pending further study of the nature of the phenomenon, but its use has been imposed and it has become an international scientific vocabulary. The rectilinear shape suggested rays that spread in a straight line, which is why Goldstein gave them the name of channel rays. If the gas contained in the tube was air, the stripes were yellow. 
Goldstein observed that, when an electric discharge was produced in a tube containing a rarefied gas, using as a negative electrode (cathode) a metal sheet normal to the axis of the tube and provided with small holes, bright rectilinear lines directed towards to the part opposite to that occupied by the positive electrode (anode). 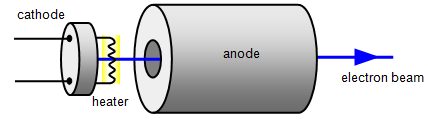
With the support of the German Academy of Sciences, he carried out numerous experiments on electric discharges in a vacuum that led to the discovery of canal rays, which he first reported to the Berlin Academy in 1886, and continued his research on the same theme until about 1915. He worked at the Potsdam Observatory (Germany) where, in 1927, he became head of the Astrophysics section, he collaborated with the Berlin Observatory (Germany) from 1878 to 1890 and from 1888 he was professor of physics at the University of Berlin. September 5, 1850, Gliwice (Kingdom of Prussia, now Poland) – December 26, 1930, Berlin (Weimar Republic, now Germany)
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
